Company News
EU MDR Compliance Trends for First Aid Kit Manufacturers in 2026
The EU Medical Device Regulation (EU MDR 2017/745) is continuously reshaping the regulatory system for medical related products in Europe, including first aid kits and emergency medical supplies. After entering 2026, the EU market will further refine and enforce compliance requirements, especially ......
Read More“Annual CE Audit Training Successfully Conducted at Yiwu Kebon Healthcare Co., Ltd.”
As global regulations continue to evolve, CE compliance has become a critical requirement for first aid kit manufacturers supplying to European markets. Recently, Yiwu Kebon Healthcare Co., Ltd. successfully conducted its annual CE audit training, reinforcing its commitment to product safety, regula......
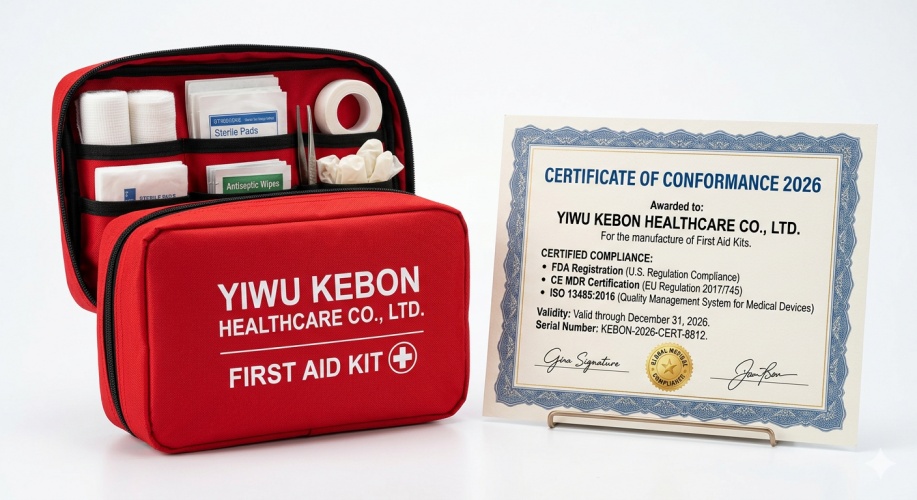
Read MoreYiwu Kebon Healthcare: 2026 FDA, CE, and ISO 13485 Certifications Renewed for First Aid Kits
As a leading manufacturer, Yiwu Kebon Healthcare announces that our First Aid Kits have officially cleared the 2026 FDA registration, CE MDR compliance, and ISO 13485 audits, ensuring medical-grade safety for global distributors
Read More